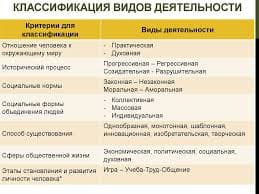
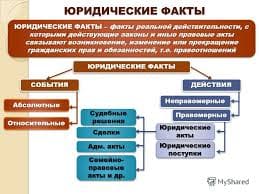
Task: 1) read the texts
2) answer the questions
3) make up a report about any famous chemist you like
Antoine Lavoisier.
Antoine Laurent Lavoisier is a French chemist, was the founder of modern chemistry.
Lavoisier carefully measured the weights of substances involved in chemical reactions. In 1772 he began a series of experiments that demonstrated the nature of combustion. He concluded that combustion results from the union of a flammable material with a newly discovered gas, which he called oxygen. Lavoisier published his findings in his Elementary Treatise on Chemistry (1789).
With French astronomer and mathematician Pierre Simon Laplace, Lavoisier conducted experiments on respiration in animals. Their studies demonstrated a similarity between common chemical reactions and the processes that occur in living organisms. These experiments provided the foundation for the science now known as biochemistry. Lavoisier also helped to develop a system for naming chemical substances based on their composition. This system is still in use.
Lavoisier was born in Paris. He received an excellent education and developed an interest in all branches of science, especially chemistry. He was elected to the French Academy of Sciences in 1768.
Lavoisier was arrested in 1793 by the leaders of the French Revolution. Many years earlier, he had become a partner in a firm that collected a number of taxes for the government. In spite of his achievements, Lavoisier was found guilty of conspiracy with the enemies of France because of his involvement in tax collection. He was executed by guillotine.
Questions
1) What famous scientist did Lavoisier work with?
2) What experiments did they conduct?
3) The foundation of what science did their experiments provide?
4) Why was he arrested?
5) What series of experiments did Lavoisier begin in 1772?
Alfred Nobel.
Alfred Bernard Nobel, a Swedish chemist, invented dynamite and founded the Nobel Prizes. As a young man, Nobel experimented with nitroglycerin in his father’s factory. He hoped to make this dangerous substance into a safe and useful explosive. He prepared a nitroglycerin explosive, but so many accidents occurred when it was put on the market that for a number of years many people considered Nobel almost a public enemy.
Finally in 1867 Nobel combined niter with an absorbent substance. This explosive could be handled and shipped safely. Nobel named it dynamite. Within a few years he became one of the world’s richest men. He set up factories throughout the world and bought the large Bofors armament plant in Sweden. He worked on synthetic rubber, artificial silk and many other products.
Nobel was never in good health. In later years he became increasingly ill and nervous. He suffered from a feeling of guilt at having created a substance that caused so much death and injury. He hated the thought that dynamite could be used in war when he had invented it for peace. Nobel set up a fund of about 9 million U.S. dollars. The interest from the fund was to be used to award annual prizes, one of which was for the most effective work in promoting international peace.
Alfred Nobel was born on October, 21, 1833 in Stockholm. He was the son of an inventor. He was educated in St. Petersburg, Russia, and later studied engineering in the United States.
Questions
1) Who was Nobel’s father?
2) What was Nobel’s chief invention?
3) Why did people consider him a public enemy for a number of years?
4) What kind of Prizes did he set up?
5) What was the interest from this fund?
Final test.
I. Match the words.
| 1) Solvent 2) depositing 3) alkali 4) particle 5) mercury 6) dioxide 7) dyestuff 8) soluble 9) charge 10) equation | a) частица b) двуокись c) заряд d) растворитель e) растворимый f) осаждение g) уравнение h) краситель i) ртуть j) щелочь |
II. Fill in the gaps with suitable words given below.
1) Natural gas is an... gas that occurs in the earth's crust.
2)... is a process used to make raw rubber useful.
3) Iron is always present as an... in commercial aluminium and its alloys.
4) Radioactivity is the..., exhibited by certain types of matter, of emitting
energy spontaneously.
5) A measure of the strength of the electrolyte is its ability... an electric current.
6) When chemical substances have the same molecular weight but different
properties, they are called....
7) Natural rubber is an elastic substance found as milky dispersion in many
species of....
8) Cellulose is a naturally occurring....
9)... is the linking of small molecules (monomers) to make large molecules.
10)... is a chemical substance which contains plant food.
Polymerization, to conduct, vulcanization, isomers, inflammable, property, plants, impurity, a fertilizer, carbohydrate.
III. Make up sentences out of these words and translate the sentences into
Russian.
1) Enough, analysis, when, is, is, the, sampling, complete, permit, subdivision,
small, to.
2) The, main, production, use, energy, fuel, of, of, is, in, atomic, uranium, a, as,
the.
3) Dealing, electrolytic, theory, solution, is, a, with, of, in, theory, compounds,
dissociation, behavior.
4) Solution, method, metals, is, depositing, from, of, a, electrolysis.
5) Composition, of, depend, only, quantitative, not, their, substances, properties,
on, qualitative, and.
IV. Make up 5 sentences of your own with the words from task I.
Литература
1. Андреев Г.Я., Гураль Л.Л., Лев А.Л. Сборник технических текстов на английском языке. М.: Издательство «Высшая школа», 1972.
2. Иллюстрированный словарь английского и русского языка с указателями. М.: Живой язык, 2003.
3. Парахина А.В. Пособие по переводу технических текстов с английского языка на русский. М.: Издательство «Высшая школа», 1972.
 2020-05-21
2020-05-21 405
405